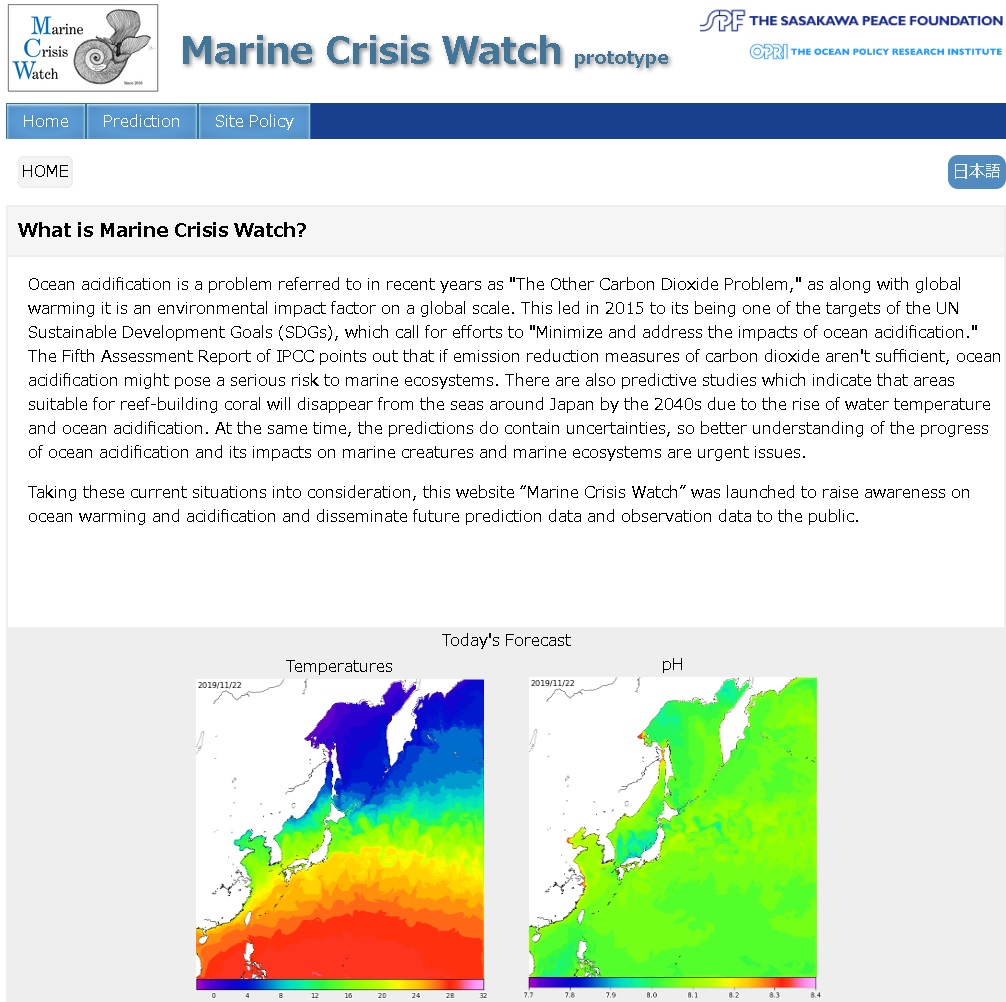
What is ocean acidification and why is it occurring?
Mr. Tsunoda: In short, ocean acidification is the process whereby carbon dioxide dissolves in the ocean and makes the water more acidic. Seawater is normally slightly alkaline on the pH scale, but as more carbon dioxide is dissolved in the oceans, the pH moves toward neutrality, or in other words, becomes more acidic. The more carbon dioxide in the air, the more carbon dioxide dissolves into the sea. Ocean acidification, like global warming, is caused by the carbon dioxide that is produced by human activities.
What are the effects of ocean acidification?Mr. Tsunoda: When seawater becomes more acidic, the number of carbonate ions in the water goes down. This creates a problem for organisms that have a shell or a skeleton made of calcium carbonate.
For example, between 2005 and 2009, ocean acidification on the west coast of the United States caused problems for oyster farming. In the Arctic Ocean and elsewhere, there are concerns about the decrease in the number of organisms with calcium carbonate shells and the subsequent effects on the marine life that feeds on those organisms. When the natural food chain breaks down, it eventually reaches human beings at the top of the chain, but it’s difficult to predict the nature of these effects or how far reaching they will be.
Item 3 of Sustainable Development Goal 14, which was announced in 2015, relates to ocean acidification. OPRI has been conducting research on acidification since 2015, and we’ve found that ocean acidification is a complex issue.